-
 Saudi's new national carrier gets off ground despite war, delays
Saudi's new national carrier gets off ground despite war, delays
-
Eddie Jones eyes Mourinho-like laundry stunt to escape ban

-
 Bollywood's Imtiaz Ali bets on Gen Z thirst for love
Bollywood's Imtiaz Ali bets on Gen Z thirst for love
-
Messi plushies see roaring trade as China firms get World Cup boost

-
 Messi sparkles on return as Somali referee says World Cup dream over
Messi sparkles on return as Somali referee says World Cup dream over
-
Iran, US trade blows as Middle East peace deal draws no nearer

-
 Salt: integral ingredient of sumo stars' art
Salt: integral ingredient of sumo stars' art
-
Staal shines as Carolina beat Vegas 5-3 to level Stanley Cup Final

-
 Messi scores on injury return as Argentina beat Iceland in World Cup warm-up
Messi scores on injury return as Argentina beat Iceland in World Cup warm-up
-
Art, maths and killing: Ukraine drone chief's formula to stop Russia

-
 Tech leads Asia losses, oil rises as rollercoaster week rumbles on
Tech leads Asia losses, oil rises as rollercoaster week rumbles on
-
Messi set to return as Somali referee says World Cup dream over

-
 Former Wallabies skipper Wright signs for Welsh club Ospreys
Former Wallabies skipper Wright signs for Welsh club Ospreys
-
Pope to bless Barcelona's Sagrada Familia, world's tallest church

-
 Emotional World Cup return to Mexico for South Africa coach Broos
Emotional World Cup return to Mexico for South Africa coach Broos
-
Bill Gates faces questioning in US Congress over Epstein ties

-
 'The Donald of Dubai': property tycoon seeks to become data king
'The Donald of Dubai': property tycoon seeks to become data king
-
PGA Tour to co-sanction Australian Open in global push

-
 Elon Musk, after DOGE and politics, bets on SpaceX IPO
Elon Musk, after DOGE and politics, bets on SpaceX IPO
-
Saudis in World Cup spotlight after $2bn spending spree

-
 Mexico doubles down on security before 2026 World Cup
Mexico doubles down on security before 2026 World Cup
-
US must not be 'too honest' at World Cup, says Roldan

-
 Italian astronaut to pilot Artemis III mission
Italian astronaut to pilot Artemis III mission
-
North Korea says Xi's visit produced 'far-reaching blueprint' for ties

-
 Benfica say farewell to Mourinho as Real Madrid return nears
Benfica say farewell to Mourinho as Real Madrid return nears
-
Protesters torch buildings and vehicles, block roads over Belfast stabbing

-
 US strikes Iran after Apache helicopter downing
US strikes Iran after Apache helicopter downing
-
Threats to US lawmakers spiked after Meta eased moderation: watchdog

-
 Nick Reiner seeks trust fund money for parent murder defense
Nick Reiner seeks trust fund money for parent murder defense
-
Spain, France qualify for 2027 Women's World Cup as England wait

-
 Protesters torch building and vehicles, block roads over Belfast stabbing
Protesters torch building and vehicles, block roads over Belfast stabbing
-
A woman in charge of the UN? Candidates feel it's about time

-
 US tech shares resume sell-off while oil prices retreat
US tech shares resume sell-off while oil prices retreat
-
Protesters block road to Mexican World Cup stadium

-
 White House World Cup chief defends visa ban for Somali referee, Iranians
White House World Cup chief defends visa ban for Somali referee, Iranians
-
Serena back in the groove on triumphant return to tennis

-
 'It doesn't matter': US star Reyna looks past World Cup scandal
'It doesn't matter': US star Reyna looks past World Cup scandal
-
Somali referee says World Cup 'dream' ruined

-
 Knicks ready to 'throw the first punch' in NBA Finals
Knicks ready to 'throw the first punch' in NBA Finals
-
'Beaten to death': the grim toll of Ecuador's security crackdown

-
 Anthropic opens most powerful AI model to public with safeguards
Anthropic opens most powerful AI model to public with safeguards
-
Serena Williams makes winning return in Queen's Club doubles

-
 Trump vows response after Iran shoots down US helicopter
Trump vows response after Iran shoots down US helicopter
-
Real Madrid's 150 mn euros bid for Atletico's Alvarez rejected

-
 Spurs handling physicality of Knicks and New York hostility
Spurs handling physicality of Knicks and New York hostility
-
Peru election chief tells AFP count could take two weeks

-
 Stokes considering England captaincy future after nightclub incident
Stokes considering England captaincy future after nightclub incident
-
Atalanta sack coach Palladino with Sarri set to arrive

-
 Italian Luca Parmitano to be first European to join an Artemis mission: NASA
Italian Luca Parmitano to be first European to join an Artemis mission: NASA
-
One killed as Kenyan protests at US Ebola centre turn violent


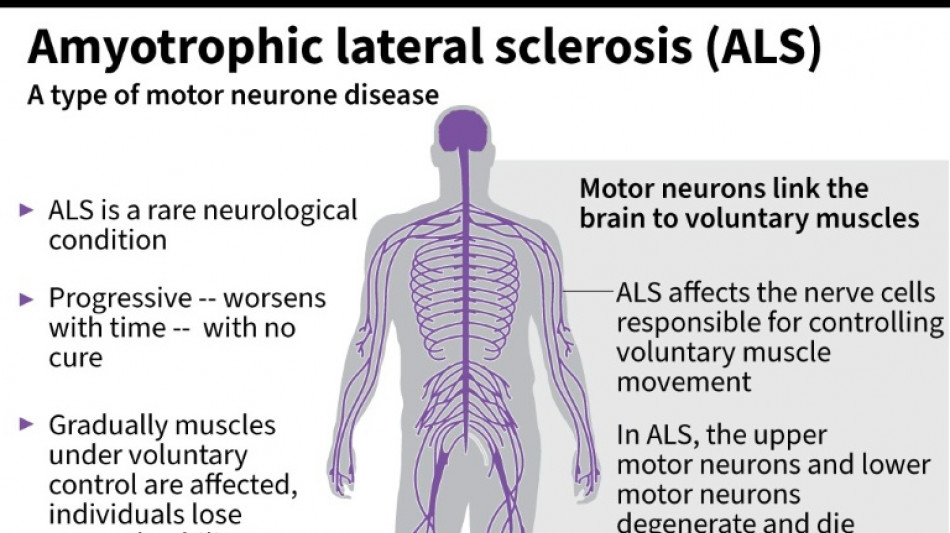
US company withdraws ALS drug after it fails in trial
Amylyx Pharmaceuticals announced Thursday it was withdrawing its approved treatment against the deadly neurodegenerative disease ALS after clinical data found no evidence the drug worked.
In a statement, the US company said it would discontinue its market authorizations for Relyvrio/Albrioza, using the brand names of the medicine in the US and Canadian markets.
"While this is a difficult moment for the ALS community, we reached this path forward in partnership with the stakeholders who will be impacted and in line with our steadfast commitment to people living with ALS and other neurodegenerative diseases," said the company's co-CEOs Joshua Cohen and Justin Klee in a statement.
The company also said it was reducing its workforce "by approximately 70 percent" as it focused on another experimental drug for use against ALS, and on repurposing Relyvrio for other conditions. It added it would continue to make Relyvrio available for patients who wish to keep using the treatment, through a "free drug program."
The news follows data from a clinical trial of 664 ALS patients announced in March, which found no significant differences in outcomes between those on the treatment group and those who received a placebo.
It was a big blow for patients with amyotrophic lateral sclerosis, sometimes called Lou Gehrig's disease after the famous baseball player, which devastates nerve cells in the brain and spinal cord.
ALS affects about two people per 100,000 every year, causing progressive loss of motor and cognitive function. Most patients die within five years of their diagnosis.
Relyvrio's approval by the US Food and Drug Administration in 2022 was controversial and based on the results of a single trial that involved just 137 participants.
The FDA itself noted there was "residual uncertainty about the evidence of effectiveness" -- but "given the serious and life-threatening nature of ALS and the substantial unmet need, this level of uncertainty is acceptable in this instance and consideration of these results in the context of regulatory flexibility is appropriate."
- Patient groups backed approval -
Advocacy groups also mounted a major campaign sending a petition to the FDA with tens of thousands of signatures urging approval. Once it became available, Amylyx reportedly announced an eye-watering list price of $158,000 per year in the US, drawing criticism.
Patient groups in Europe watched with desperation at the bureaucratic delays.
When the European Union drug watchdog later announced it was rejecting Relyvrio, the decision was slammed as "an affront" by angry French patients, who say they "don't have time to wait." France later relented, offering conditional approval in November.
"We commend Amylyx for pulling Relyvrio off the market, while still ensuring that people living with ALS can access the drug if they believe it is helping them," said the US-based ALS association, which had lobbied for the drug's approval and funded its research.
"Safe and potentially effective treatments can be made accessible rapidly until further research can confirm their efficacy," it added.
For now, there remain only a handful of treatments available.
Riluzole, FDA approved in 1995, prolongs life approximately three months. Edaravone, FDA approved in 2017, has been found to slow disease progression and improve survival.
And in 2023, the regulatory body approved tofersen, a gene therapy treatment that targets those ALS cases that are caused by mutations in the SOD1 gene.
H.Nasr--SF-PST




