-
 Xiaomi quarterly profit slumps despite annual EV gains
Xiaomi quarterly profit slumps despite annual EV gains
-
Iran, Israel trade strikes despite Trump talk of negotiations

-
 IPL's Bengaluru to keep 11 seats empty in honour of stampede dead
IPL's Bengaluru to keep 11 seats empty in honour of stampede dead
-
Oil prices jump, stocks waver after Trump's Iran claim

-
 'A top person': Who is the US dealing with in Iran?
'A top person': Who is the US dealing with in Iran?
-
In Lebanon's Tyre, ancient site threatened by Israeli bombs

-
 US-Israeli war on Iran is 'breach of international law': German president
US-Israeli war on Iran is 'breach of international law': German president
-
Iran strikes Israel, denies Trump talks

-
 Mbappe says injury is behind him, all systems go for World Cup
Mbappe says injury is behind him, all systems go for World Cup
-
Supporters' group file lawsuit against 'excessive' World Cup ticket prices

-
 Gas shortages push India's poor back to wood and coal
Gas shortages push India's poor back to wood and coal
-
'Plundered': Senegal fishers feel sting of illegal, industrial vessels

-
 Iran hits Israel with missiles after denying Trump talks
Iran hits Israel with missiles after denying Trump talks
-
Stocks rise on Trump U-turn but unease sees oil bounce

-
 Trans community alarmed as India moves to curb LGBTQ rights
Trans community alarmed as India moves to curb LGBTQ rights
-
Families' nightmare fight for justice in Austria child sex cases

-
 Tiger Woods to return to action in TGL with Masters looming
Tiger Woods to return to action in TGL with Masters looming
-
Australia, EU agree sweeping new trade pact eight years in the works

-
 Back to black: facing energy shock, Asia turns to coal
Back to black: facing energy shock, Asia turns to coal
-
Iran fires new wave of missiles at Israel after denying Trump talks

-
 Manila's jeepney drivers struggle as Mideast war sends diesel cost soaring
Manila's jeepney drivers struggle as Mideast war sends diesel cost soaring
-
The contenders vying to be next Danish leader

-
 India's historic haveli homes caught between revival and ruin
India's historic haveli homes caught between revival and ruin
-
Denmark votes in close election, outgoing PM tipped to win

-
 N. Korea's Kim vows 'irreversible' nuclear status, warns Seoul of 'merciless' response
N. Korea's Kim vows 'irreversible' nuclear status, warns Seoul of 'merciless' response
-
Pressure on Italy as play-off hopefuls eye 2026 World Cup

-
 Malinin and Sakamoto seek solace at figure skating worlds as Olympic champions absent
Malinin and Sakamoto seek solace at figure skating worlds as Olympic champions absent
-
'Perfect Japan' posts spark Gen Z social media backlash

-
 Asian stocks rise on Trump U-turn but unease sees oil bounce
Asian stocks rise on Trump U-turn but unease sees oil bounce
-
Pistons halt Lakers streak while Spurs, Thunder win

-
 Silence not an option, says Canadian Sikh activist after fresh threats
Silence not an option, says Canadian Sikh activist after fresh threats
-
Rennie shakes up All Blacks backroom team as 2027 World Cup looms

-
 Australia, EU agree to sweeping new trade pact after eight years
Australia, EU agree to sweeping new trade pact after eight years
-
Too old? The 92-year-old US judge handling Maduro case

-
 Australia, EU agree sweeping new trade pact
Australia, EU agree sweeping new trade pact
-
Sinner, Sabalenka march on in Miami as more seeds crash out

-
 US social media addiction trial jury struggles for consensus
US social media addiction trial jury struggles for consensus
-
EU 'concerned' by reports Hungary leaked information to Russia

-
 USPA Global and ESPN Expand Relationship with Chris Fowler for 2026 High-Goal Polo Championships
USPA Global and ESPN Expand Relationship with Chris Fowler for 2026 High-Goal Polo Championships
-
EU chief meets Australian PM as trade talks enter 'last mile'

-
 Israel pounds south Beirut, says captured Hezbollah members
Israel pounds south Beirut, says captured Hezbollah members
-
EU chief to meet Australian PM as trade talks enter 'last mile'

-
 Champion Mensik, Medvedev dumped out of Miami Open
Champion Mensik, Medvedev dumped out of Miami Open
-
Jury at US social media addiction trial reports 'difficulty' in finding consensus

-
 Stokes eager to lead England recovery after 'hardest period of captaincy'
Stokes eager to lead England recovery after 'hardest period of captaincy'
-
Venezuela protesters demand end to 'hunger' level wages

-
 Eight people arrested in Brazil for 'brutal' attack on capybara
Eight people arrested in Brazil for 'brutal' attack on capybara
-
Audi Q9 – how likely is it to become a reality?

-
 Oil slides, stocks rebound on Trump's Iran remarks
Oil slides, stocks rebound on Trump's Iran remarks
-
On Iran, Trump executes his most spectacular U-turn yet


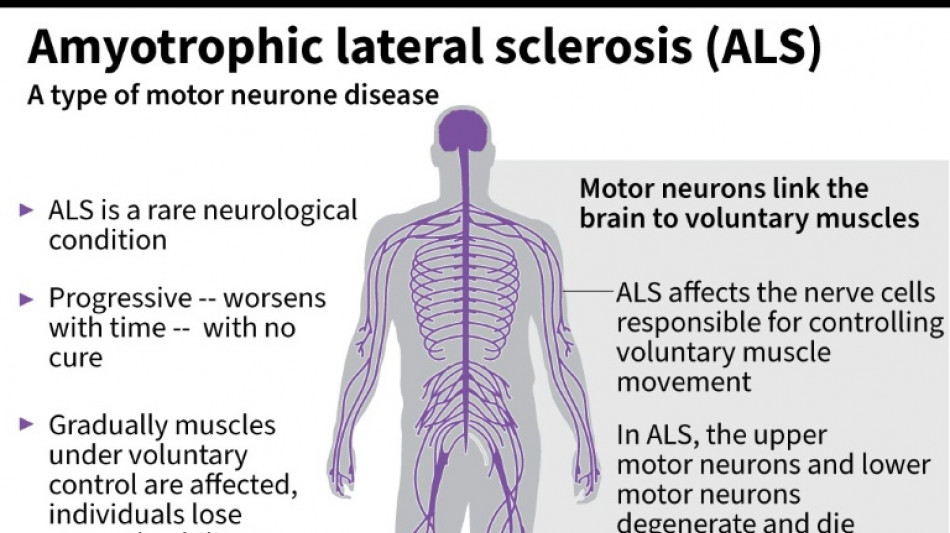
Every month counts: European ALS patients want new drugs
Olivier Goy is running out of time.
The French entrepreneur was diagnosed in 2020 with amyotrophic lateral sclerosis (ALS) -- the incurable neurodegenerative disease that normally claims the lives of patients within three to five years.
There are new treatments that have given patients hope of being able to extend their lives by an invaluable few months, but the approval process in Europe is taking time, infuriating desperate patients.
"When you are certain to die soon, patients and some doctors are ready to take some risks," Goy told AFP.
In response to the lack of new treatments in his native France, the founder of the fintech start-up October spends 3,000 euros ($3,180) every month to buy the ingredients to make his own drugs.
ALS, also known as Lou Gehrig's disease, attacks the motor nerve cells in the brain and spinal cord, progressively paralysing muscles until patients cannot walk, eat, speak or breathe.
Around one in 10,000 people have the disease in the EU, according to the European Medicines Agency.
The drug Riluzole, which has been available in Europe and the UK since the 1990s, is capable of prolonging the lives of patients by around three months.
But otherwise, no new treatment has been approved in Europe for more than two decades.
- 'First hope in 20 years' -
A new treatment called AMX0035 was given the green light in the United States and Canada last year.
"It is the first hope we have had in 20 years: the first drug which is aimed at everyone and which had results" suggesting up to six months in added life expectancy, said Sabine Turgeman, head of the French Association for Research into ALS.
But the extent of the benefits of AMX0035 remains unclear. The US Food and Drug Administration approved the drug, sold under the name Relyvrio, based on the results of a single Phase 2 trial that involved just 137 participants.
The drug's developer, Amylyx Pharmaceuticals, is conducting larger, more comprehensive trials, with results expected in 2024.
Amylyx said earlier this month that the European Union's drug watchdog EMA is reviewing its submission for approval and it expects a decision in the first half of this year.
But for those with the disease, every delay represents a significant amount of the time they have left.
"It's not going fast enough," Turgeman said. "This disease is not on bureaucratic time".
For European patients who cannot afford to import their own ingredients like Goy, the only way to get access to new treatments is to join a clinical trial.
But such trials have very specific criteria for selection -- and even if a patient gets in, there is a chance they will be in the group given a placebo.
- 'Totally abandoned' -
Given how swiftly the disease progresses, patients and families are pressing for more options.
"We feel totally abandoned," said Sophie Garofalo, whose brother was diagnosed with ALS five years ago.
His family tried to enter him into clinical trials, "but either he does not meet the criteria, or the trials have already started," she said.
"He is ready to take anything, try everything".
French pharmaceutical company AB Science is developing another potential treatment using the drug masitinib, which initial results suggest could add months to the lives of patients.
The firm's CEO Alain Moussy said that because "time is very limited" for ALS patients, there should be more flexibility in the approval system.
"What degree of risk should be taken? That's for the health agencies to answer -- but they can guided by policymakers and patients," he said.
A.AlHaj--SF-PST



