-
 Pistons rout Magic to complete comeback, advance in NBA playoffs
Pistons rout Magic to complete comeback, advance in NBA playoffs
-
Trump says US and Iran in 'positive' talks, unveils plan to escort Hormuz ships

-
 Talisman Endrick fires resurgent Lyon into third in France
Talisman Endrick fires resurgent Lyon into third in France
-
Verstappen laments spin and struggle for pace in Miami

-
 Teen Antonelli wins again in Miami to extend title race lead
Teen Antonelli wins again in Miami to extend title race lead
-
Ferrari's Leclerc admits he threw away Miami podium finish

-
 Cristian Chivu, a winner with Inter on the pitch and in the dugout
Cristian Chivu, a winner with Inter on the pitch and in the dugout
-
Key players from Inter Milan's Serie A title triumph

-
 No.4 Young cruises to PGA title at Doral
No.4 Young cruises to PGA title at Doral
-
Vinicius double delays Barca title as Real Madrid down Espanyol

-
 Inter Milan win Italian title for third time in six seasons
Inter Milan win Italian title for third time in six seasons
-
Spurs solved mental frailty to boost survival bid: De Zerbi

-
 Miami champ Antonelli shrugs off success, vows 'back to work'
Miami champ Antonelli shrugs off success, vows 'back to work'
-
Man Utd beat Liverpool, Spurs climb out of relegation zone

-
 Spurs out of relegation zone after vital win at Villa
Spurs out of relegation zone after vital win at Villa
-
No.1 Korda cruises to LPGA Mexico crown

-
 Thompson-Herah shines at world relays, Tebogo helps Botswana to win
Thompson-Herah shines at world relays, Tebogo helps Botswana to win
-
Three die on Atlantic cruise ship from suspected hantavirus: WHO

-
 Germany's Merz says not 'giving up on working with Donald Trump'
Germany's Merz says not 'giving up on working with Donald Trump'
-
Mercedes' Kimi Antonelli wins Miami Grand Prix

-
 Man Utd job feels 'natural' to Carrick
Man Utd job feels 'natural' to Carrick
-
Ferguson taken to hospital before Man Utd win against Liverpool

-
 'Devil Wears Prada 2' takes top spot in N. America box office
'Devil Wears Prada 2' takes top spot in N. America box office
-
Iran weighs US response to peace plan after warning against military action

-
 Gladbach sink Dortmund, St Pauli edge closer to drop
Gladbach sink Dortmund, St Pauli edge closer to drop
-
Rubio to visit Rome, meet Pope Leo after Trump row
-
 Kyiv hits Russian oil sites as eight killed in both countries
Kyiv hits Russian oil sites as eight killed in both countries
-
Iran says US military operation 'impossible' as Trump mulls peace proposal

-
 Man Utd beat Liverpool to secure Champions League place
Man Utd beat Liverpool to secure Champions League place
-
Two die in 'respiratory illness' outbreak on Atlantic cruise ship

-
 Barcelona sink Bayern to reach women's Champions League final
Barcelona sink Bayern to reach women's Champions League final
-
True Love lands eighth English 1000 Guineas for O'Brien

-
 Sinner dismantles Zverev to win Madrid Open, set record
Sinner dismantles Zverev to win Madrid Open, set record
-
Brilliant Bordeaux clean out Bath to reach Champions Cup final

-
 Second unexploded shell found at illegal French rave: minister
Second unexploded shell found at illegal French rave: minister
-
Bournemouth eye European place after crushing Palace

-
 Pogacar ends dominant Tour of Romandie with fourth win
Pogacar ends dominant Tour of Romandie with fourth win
-
Chakravarthy, Narine help Kolkata stay alive in IPL

-
 Daughter says Maradona died after carers' plan 'went out of control'
Daughter says Maradona died after carers' plan 'went out of control'
-
OPEC+ hikes oil production quotas but stays mum on UAE pull-out

-
 Two women suffocate on migrant boat seeking to reach UK
Two women suffocate on migrant boat seeking to reach UK
-
How Schalke returned to the Bundesliga after their 'worst season ever'

-
 Two women die on migrant boat seeking to reach UK
Two women die on migrant boat seeking to reach UK
-
Mumbai coach Jayawardene backs Suryakumar to find his 'rhythm'

-
 Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach
Under full moon, Shakira thrills 2 million fans on Rio's Copacabana beach
-
Bangkok food vendor curbs push city staple from the streets

-
 More Nepalis drive electric, evading global fuel shocks
More Nepalis drive electric, evading global fuel shocks
-
Latecomer Japan eyes slice of rising global defence spending

-
 Messi goal not enough as Miami collapse in 4-3 loss to Orlando
Messi goal not enough as Miami collapse in 4-3 loss to Orlando
-
German fertiliser makers and farmers struggle with Iran war fallout


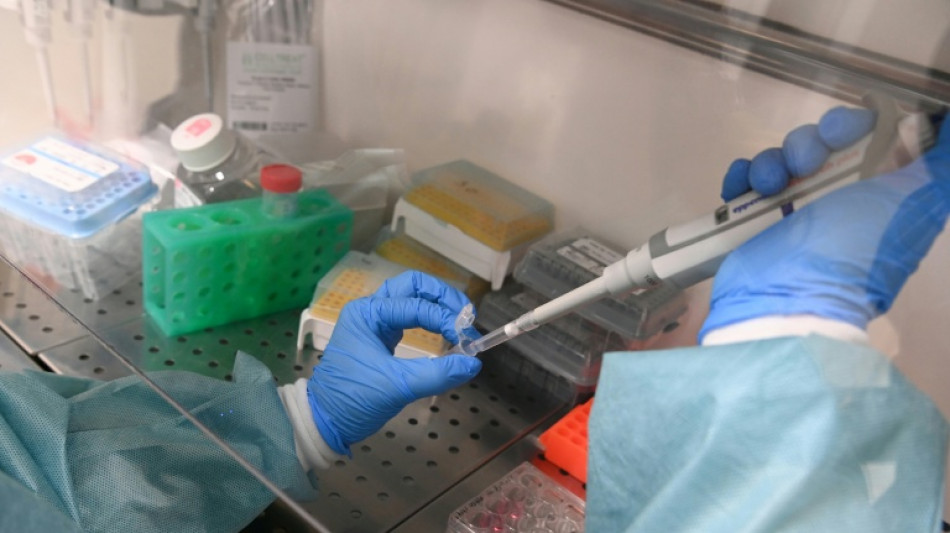
Moderna says US refusing to review mRNA-based flu shot
Vaccine manufacturer Moderna said Tuesday the US Food and Drug Administration was refusing to review an application for its first mRNA-based flu shot.
The move comes as the current iteration of the FDA has called for a reconsideration of approval procedures for certain vaccines, including for influenza -- proposed federal policy changes under President Donald Trump that have triggered widespread alarm among public health and medical professionals.
Moderna, a US company, said the agency's top vaccine regulator, Vinay Prasad, wrote in a letter that Moderna's clinical trial was not "adequate and well-controlled," and had not tested its experimental shot against the best product on the market.
In the large trial Moderna had compared its new vaccine with Fluarix, an approved flu shot from the company GSK.
Moderna said the rejection was "inconsistent with previous written communications" with the FDA branch that regulates biological products, including vaccines, known by the acronym CBER.
The decision "did not identify any safety or efficacy concerns with our product" and "does not further our shared goal of enhancing America's leadership in developing innovative medicines," Moderna CEO Stephane Bancel said in the statement.
"It should not be controversial to conduct a comprehensive review of a flu vaccine submission that uses an FDA-approved vaccine as a comparator in a study that was discussed and agreed on with CBER prior to starting."
Moderna said it had received a Refusal-to-File letter, which would indicate the application did not meet the requirements for substantive review. The company said it had requested a meeting to discuss the decision.
The letter did not identify any safety or efficacy concerns regarding the mRNA vaccine, said Moderna, which added the shot was accepted for review in the European Union, Canada and Australia.
During his first term Trump called mRNA technology a "modern-day miracle."
It was used during the Covid-19 pandemic to swiftly develop an immunization against the fast-spreading illness, and was credited with saving millions of lives.
Vaccines against Covid-19 are considered safe and effective by global health authorities, and protect against the most severe forms of the infection.
But in his second term, Trump has appointed Robert F. Kennedy Jr as his health chief, and the long-time vocal vaccine skeptic has spent the past year reshaping federal health agencies in his image.
That has meant spreading false information and sowing doubt about the safety and efficacy of vaccines widely known to be safe, and upending the pediatric immunization schedule.
Notably, Kennedy cut off federal research grants that funded mRNA development.
X.Habash--SF-PST
