-
 Iran fires new wave of missiles at Israel after denying Trump talks
Iran fires new wave of missiles at Israel after denying Trump talks
-
Manila's jeepney drivers struggle as Mideast war sends diesel cost soaring

-
 The contenders vying to be next Danish leader
The contenders vying to be next Danish leader
-
India's historic haveli homes caught between revival and ruin

-
 Denmark votes in close election, outgoing PM tipped to win
Denmark votes in close election, outgoing PM tipped to win
-
N. Korea's Kim vows 'irreversible' nuclear status, warns Seoul of 'merciless' response

-
 Pressure on Italy as play-off hopefuls eye 2026 World Cup
Pressure on Italy as play-off hopefuls eye 2026 World Cup
-
Malinin and Sakamoto seek solace at figure skating worlds as Olympic champions absent

-
 'Perfect Japan' posts spark Gen Z social media backlash
'Perfect Japan' posts spark Gen Z social media backlash
-
Asian stocks rise on Trump U-turn but unease sees oil bounce

-
 Pistons halt Lakers streak while Spurs, Thunder win
Pistons halt Lakers streak while Spurs, Thunder win
-
Silence not an option, says Canadian Sikh activist after fresh threats

-
 Rennie shakes up All Blacks backroom team as 2027 World Cup looms
Rennie shakes up All Blacks backroom team as 2027 World Cup looms
-
Australia, EU agree to sweeping new trade pact after eight years

-
 Too old? The 92-year-old US judge handling Maduro case
Too old? The 92-year-old US judge handling Maduro case
-
Australia, EU agree sweeping new trade pact

-
 Sinner, Sabalenka march on in Miami as more seeds crash out
Sinner, Sabalenka march on in Miami as more seeds crash out
-
US social media addiction trial jury struggles for consensus

-
 EU 'concerned' by reports Hungary leaked information to Russia
EU 'concerned' by reports Hungary leaked information to Russia
-
EU chief meets Australian PM as trade talks enter 'last mile'

-
 Israel pounds south Beirut, says captured Hezbollah members
Israel pounds south Beirut, says captured Hezbollah members
-
EU chief to meet Australian PM as trade talks enter 'last mile'

-
 Champion Mensik, Medvedev dumped out of Miami Open
Champion Mensik, Medvedev dumped out of Miami Open
-
Jury at US social media addiction trial reports 'difficulty' in finding consensus

-
 Stokes eager to lead England recovery after 'hardest period of captaincy'
Stokes eager to lead England recovery after 'hardest period of captaincy'
-
Venezuela protesters demand end to 'hunger' level wages

-
 Eight people arrested in Brazil for 'brutal' attack on capybara
Eight people arrested in Brazil for 'brutal' attack on capybara
-
Audi Q9 – how likely is it to become a reality?

-
 Oil slides, stocks rebound on Trump's Iran remarks
Oil slides, stocks rebound on Trump's Iran remarks
-
On Iran, Trump executes his most spectacular U-turn yet

-
 Trump announces 'very good' Iran talks denied by Tehran
Trump announces 'very good' Iran talks denied by Tehran
-
Bill Cosby ordered to pay $19m over sex abuse claim

-
 Dodgers eye 'threepeat' as new MLB season welcomes robot umpires
Dodgers eye 'threepeat' as new MLB season welcomes robot umpires
-
Dacia Striker: Stylish and sturdy?

-
 Skoda Peaq: New all-electric seven-seater
Skoda Peaq: New all-electric seven-seater
-
Medvedev ousted by Cerundolo at Miami Open

-
 Runway collision kills two pilots at New York airport
Runway collision kills two pilots at New York airport
-
Italian voters reject Meloni's reforms in referendum blow

-
 Bosnian truckers blocked EU freight terminals for a day over visa rules
Bosnian truckers blocked EU freight terminals for a day over visa rules
-
Colombia military aircraft crashes with 125 aboard, many feared dead

-
 Rip-offs at the petrol pump?
Rip-offs at the petrol pump?
-
Shakira to wrap up world tour with Madrid residency

-
 World gave Israel 'licence to torture Palestinians': UN expert
World gave Israel 'licence to torture Palestinians': UN expert
-
Colombia says 80 troops on crashed aircraft, many feared dead

-
 France turns to 2027 race to succeed Macron
France turns to 2027 race to succeed Macron
-
New Mercedes GLC electric

-
 Namibia rejects Starlink licence request
Namibia rejects Starlink licence request
-
Ex-model questioned in France over scout with Epstein links

-
 UK sending air defence systems to Gulf: PM
UK sending air defence systems to Gulf: PM
-
Trump administration seeks to ease oil fears but industry wary


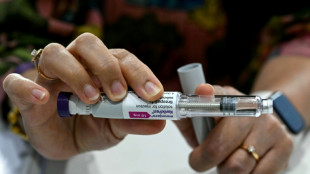
US approves new drug to treat Alzheimer's disease
The US Food and Drug Administration on Friday approved a highly anticipated new drug designed to slow cognitive decline in patients in mild and early stages of Alzheimer's disease.
The FDA approval of the drug, Leqembi, also known as lecanemab, comes just days after the regulatory agency was harshly criticized in a congressional report for its green-lighting of another Alzheimer's drug, Aduhelm.
And it was granted despite trial results showing the monoclonal antibody treatment carries risks of brain swelling and bleeding.
Both drugs were approved through an accelerated process that allows the FDA to fast-track approval of drugs for serious conditions where there is an unmet medical need.
Leqembi and Aduhelm, which were jointly developed by Japan's Eisai and Biogen of the United States, "represent an important advancement in the ongoing fight to effectively treat Alzheimer's disease," the FDA said in a statement.
"Alzheimer's disease immeasurably incapacitates the lives of those who suffer from it and has devastating effects on their loved ones," Billy Dunn of the FDA's Center for Drug Evaluation and Research said in a statement.
Leqembi, Dunn said, is "the latest therapy to target and affect the underlying disease process of Alzheimer's, instead of only treating the symptoms of the disease."
Approximately 6.5 million Americans suffer from Alzheimer's, which is characterized by memory loss and declining mental acuity.
Preliminary data from a trial of Leqembi was released in September and found it slowed cognitive decline in Alzheimer's patients by 27 percent.
The phase three trial involved nearly 1,800 people, divided between those given the drug and given a placebo, and ran over 18 months.
The complete trial data, published in the New England Journal of Medicine, raised concern about the incidence of "adverse effects" including brain bleeds and swelling.
The results showed that 17.3 percent of patients administered the drug experienced brain bleeds, compared with nine percent of those receiving a placebo.
And 12.6 percent of those taking the drug experienced brain swelling, compared with just 1.7 percent of those in the placebo group.
Deaths were reported at approximately the same rate in both arms of the trial of the drug.
- $26,500 a year -
In Alzheimer's disease, two key proteins, tau and amyloid beta, build up into tangles and plaques, known together as aggregates, which cause brain cells to die and lead to brain shrinkage.
Leqembi, which is administered intravenously once every two weeks, works by targeting amyloid.
In the trial, patients receiving Leqembi had a statistically significant reduction in brain amyloid plaque compared to the placebo arm, which had no reduction of amyloid beta plaque.
Biogen and Eisai previously brought Aduhelm to market, but there was significant controversy over whether it worked, and its approval in 2021 led to three high-level resignations in the FDA.
A US congressional investigation said the accelerated approval process for Aduhelm, the first drug approved in decades to treat Alzheimer's, was "rife with irregularities" and criticized both the agency and Biogen.
The Cambridge, Massachusetts-based Biogen set an "unjustifiably high price" for Aduhelm of $56,000 a year, the congressional report said.
Eisai said Leqembi would be priced initially at $26,500 per year and estimated that 100,000 Americans could be receiving the drug by three years from now.
Joanne Pike, president and CEO of the Alzheimer's Association, welcomed the approval of Leqembi but expressed concern that its high cost could put it out of reach of most Americans, particularly if it is not covered by Medicare, the government health insurance program for the elderly.
"People living with this fatal disease today do not have time to wait for a miracle drug or cure," Pike said in a statement.
B.Mahmoud--SF-PST